
Single Pass or Double Pass? – Making the Right Decision in Dialysis Water Purification System Selection
February 18, 2026
Hemodialysis Water Treatment System Operation and Maintenance Guideline
April 17, 2026The Critical Role of Water Quality in Modern Hemodialysis
In modern hemodialysis treatment, water quality is one of the most critical factors determining the success of the therapy. For this reason, healthcare professionals and technical teams constantly ask the same question: Why is Ultra Pure Dialysis Water Important? This question is not merely about a technical detail; it is directly related to patient safety, inflammation control, and long-term quality of life.
During hemodialysis, the patient’s blood comes into contact with the dialysate through a semi-permeable membrane. Since a large part of the dialysate consists of water, the chemical and microbiological properties of the water used directly affect the patient’s circulatory system. Therefore, the question Why is Ultra Pure Dialysis Water Important? is central to modern dialysis practices.
Considering that a patient undergoes dialysis several times a week, it is estimated that they are indirectly exposed to tens of thousands of liters of water annually. Even the smallest contamination in such a large volume of water can lead to clinical consequences.
Clinical Role and Physiological Effects of Dialysis Water

In dialysis treatment, water is not merely a carrier fluid. It directly affects electrolyte balance, toxin removal capacity, and metabolic stability. Therefore, clinicians constantly focus on the question: Why is Ultra Pure Dialysis Water Important?
When water quality is inadequate, endotoxins, heavy metals, or microbiological contaminants can indirectly reach the patient’s body via the dialysate. This can lead to acute reactions or chronic inflammation.
Scientific studies have shown that inflammatory markers are lower in dialysis centers using ultra pure water.
This clinical difference directly impacts treatment quality and leads to improved patient prognosis.
The Role of Reverse Osmosis (RO) Systems
Reverse osmosis technology forms the basis of modern water purification infrastructure. The majority of dissolved ions, heavy metals, and bacteria are removed by these systems.
Double-pass RO systems ensure that water passes through two separate membrane stages. This structure enhances microbiological safety and significantly reduces endotoxin levels.
High-efficiency RO systems not only provide chemical purification but also ensure continuous quality.
Carbon Filtration and Deionization Processes
Activated carbon filters remove oxidizing agents such as chlorine and chloramine. These substances can cause membrane damage and hemolytic reactions.
Deionization systems, on the other hand, reduce conductivity by removing dissolved ions. This process maintains the chemical stability of the water.
- Chlorine and chloramine removal
- Reduction of ion load
- Preservation of chemical stability
- Extension of membrane life
Ultrafiltration and Endotoxin Control
Ultrafilters used in the final stage minimize bacterial load. Endotoxins are cell wall components of gram-negative bacteria and can trigger inflammation even at low levels.
If endotoxin control is not ensured, pyrogenic reactions may occur. Clinical symptoms such as fever, chills, and hypotension may be observed.
Ultrafiltration minimizes microbiological risks and stabilizes treatment.
Biofilm Control in Distribution Lines
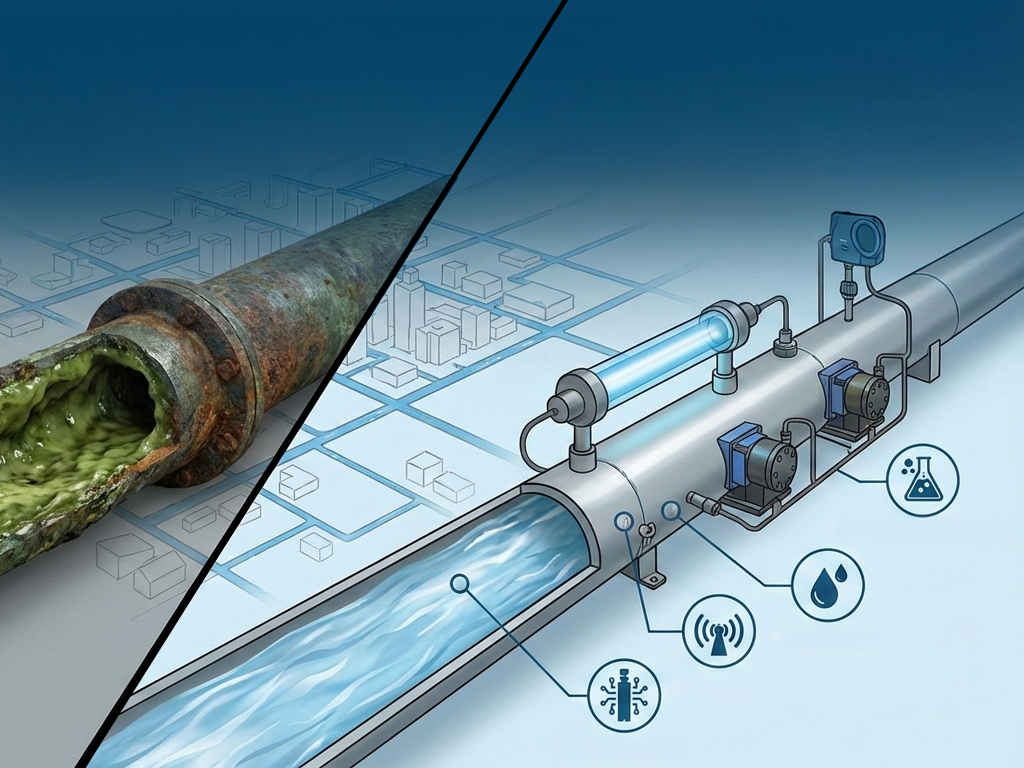
The quality of purified water does not depend solely on the production stage. Biofilm formation in distribution lines is one of the biggest sources of contamination.
Biofilm formation leads to bacterial colonization and results in increased endotoxin levels.
- Regular thermal disinfection
- Chemical cleaning protocols
- Loop design optimization
- Validation processes
Quality Control in Dialysis Centers
Safety cannot be ensured without continuity in the production of Ultra Pure Dialysis Water. Therefore, quality control processes are an integral part of the dialysis infrastructure.
Microbiological Analyses
CFU counts and endotoxin tests must be performed regularly.
Chemical Monitoring
Conductivity, pH, and heavy metal analyses must be regularly checked.
Validation and Documentation
Recording all processes is mandatory for regulatory compliance.
International Standards and Regulations
Quality criteria for Ultra Pure Dialysis Water are defined by international standards. The ISO 23500 series and AAMI criteria are referenced worldwide.
These standards define chemical and microbiological limits and ensure the protection of patient safety.
MDR and Risk Management Perspective
The European Medical Device Regulation has made risk management mandatory. Dialysis water quality is central to this risk management.
A system without a safe water infrastructure may face problems with regulatory compliance.
Impacts on Long-Term Patient Health
Low-level endotoxin exposure can lead to chronic microinflammation and increase cardiovascular risks.
In centers using ultra pure water:
- Lower inflammation
- Better anemia control
- More stable clinical outcomes
- Better quality of life
Conclusion: A Clinical Necessity
In modern hemodialysis practices, the quality of Ultra Pure Dialysis Water is no longer an option. It is an essential component of clinical safety.
Ultra pure dialysis water is indispensable for patient safety, clinical performance, and sustainable healthcare.
Your Reliable Solution Partner
Professional infrastructure setup and sustainable quality require experienced engineering support. You can establish a safe water infrastructure in your center with system design, installation, and validation services compliant with international standards. hemodialysiswaterpurification.com






